CSD-Materials in action: Pfizer. Reducing risk in solid form selection of APIs
Here we summarise a publication from our collaboration with Pfizer, which demonstrates the use of the healthcheck methodology to reduce risk and select the most stable form across three different APIs.
Summary
In this work three active pharmaceutical compounds were assessed for stability using a combination of CCDC tools, to select the most stable solid form for further development.
As well as supporting and validating lab work, in one case this “solid form health check” assessment raised the possibility of a significant weakness with an existing form that was not identified in initial experiments.
Follow up experimental work confirmed the solid form health check findings, and a new form with better characteristics was found to form in certain conditions.
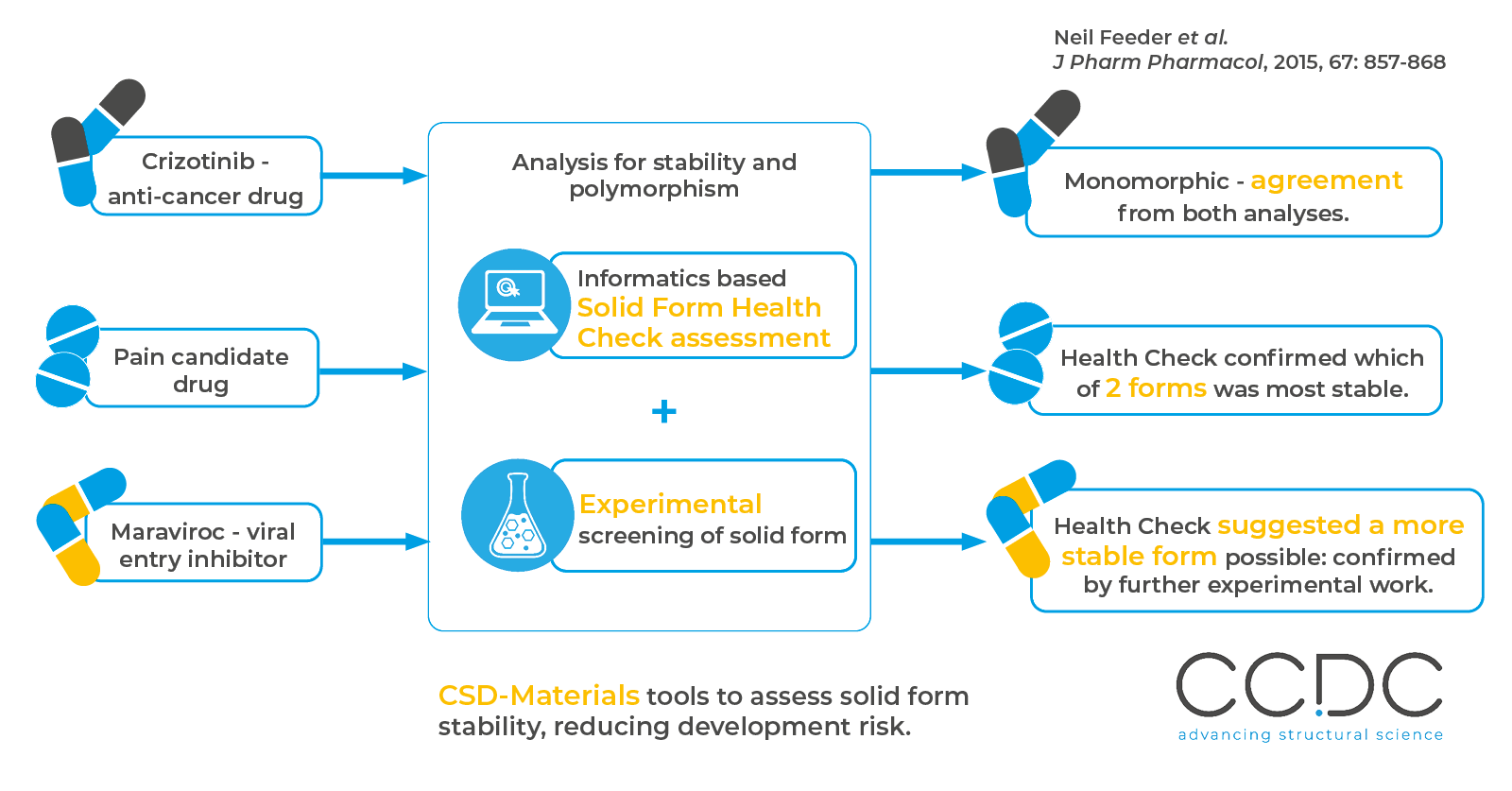
Summary of the analyses Pfizer conducted using the CCDC Solid Form Health Check assessment method. Three pharmaceuticals were evaluated, with issues found in one that were later confirmed by lab testing.
Why
Changes in conditions can produce different polymorphic forms of a crystalline material — with potentially big impacts in solubility, stability and more. In pharmaceutical development it is therefore vital the solid form is fully understood. Unforeseen polymorphs have cost drug companies hundreds of thousands of pounds in the past, for example the case of Ritonavir (Bauer et al, Pharm Res, 2001).
The “health check” assessment described in this work is a collection of in silico analyses which complement and add to experimental work already done to address this risk. By combining experimental and informatics results in this way, scientists can have greater confidence in their decision to take a structure on to further development.
How
The team employed a range of informatics analyses to fully characterise the intermolecular interactions, intramolecular geometry, chirality, Z’ and more — and coined the term “health check” to encompass this complete review of the structure. This work is done in addition to laboratory work, to strengthen and support findings there.
Across the three molecules studied here;
- Crizotinib: health check in agreement with experimental work.
- Pain candidate drug: health check confirmed which of two experimental forms was more stable.
- Maraviroc: health check suggested a more stable form could be possible. Additional lab work later confirmed this.
Read more
Read the Journal of Pharmacy and Pharmacology paper here.
Learn more about our Solid Form Health Check service, where our expert team securely assesses your structure with this method.