Tellurium
Tellurium:

Image showing Tellurium crystal
Facts about Tellurium:
- Tellurium: Tellurium is silver/white, rare, brittle, toxic
- Fun fact about Tellurium: Tellurium is one of the rarest elements on Earth, but plentiful in space. The rarity of this element is comparable to that of platinum. It is present in the earths crust in about 0.001 parts per million.
- Chemical symbol: Te
- Atomic number: 52
A crystal structure containing Tellurium:
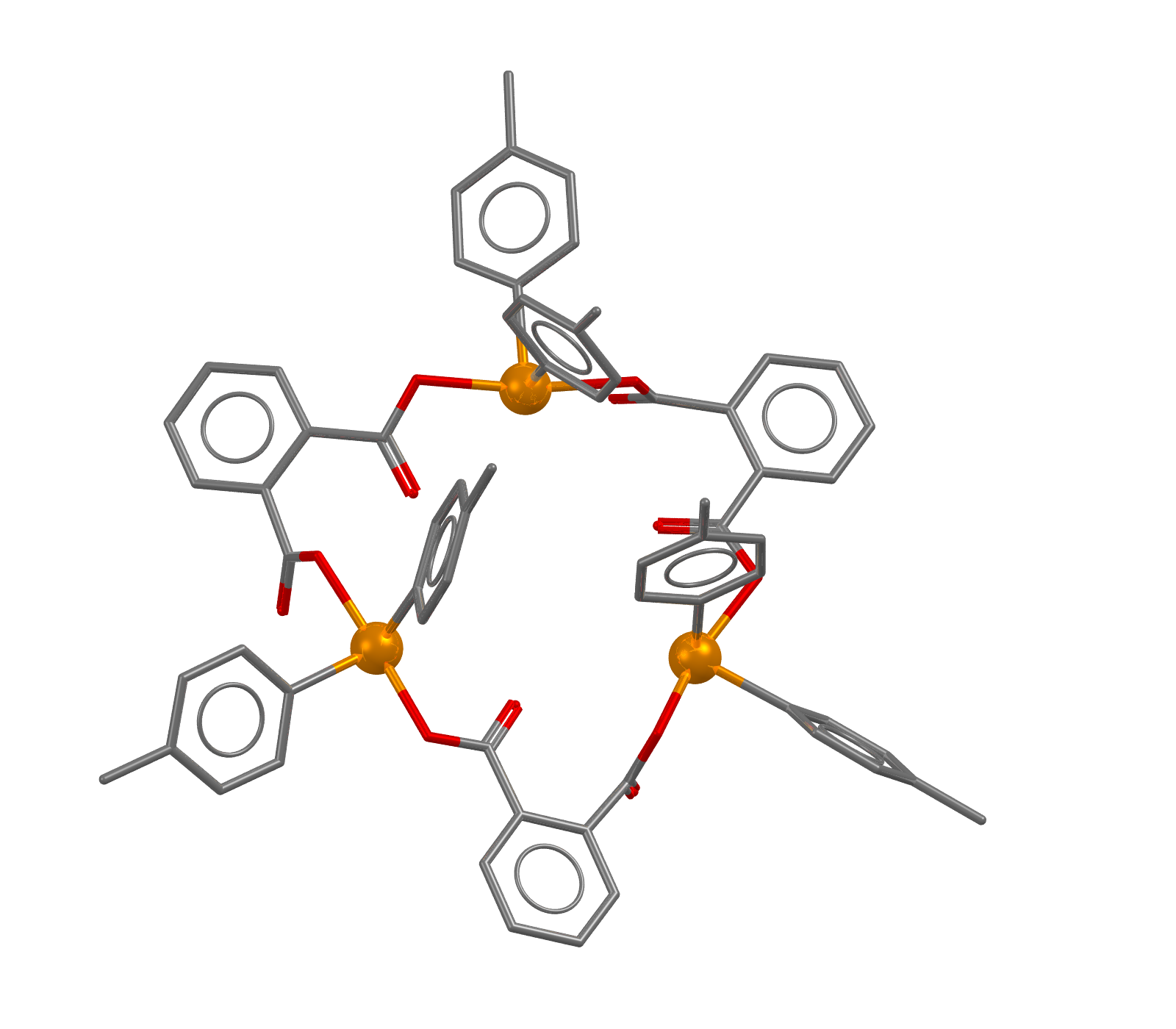
Image showing the structure of CSD Entry ACAFAL. Hydrogen atoms and solvent molecules have been omitted for clarity. Tellurium atoms are shown in orange.
Facts about this structure:
- Formula: C66 H54 O12 Te3,0.5(C8 H10),3(C2 H3 N)
- Structure name: tris((μ2-Phthalato-O,O’)-bis(p-tolyl)-tellurium) acetonitrile p-xylene solvate
- Fun fact about the structure: This structure is described as a macrocyclic multi-tellurane with hypervalent Te-O apical linkages. This structure, along with the other in the same publication were the first examples of macrocycles composed of only hypervalent apical linkages in the main chain for group 13-17 elements.
- CSD refcode: ACAFAL (What’s this?)
- Associated publication: K.Kobayashi, H.Izawa, K.Yamaguchi, E.Horn, N.Furukawa, Chemical Communications, 2001, 1428, DOI: 10.1039/b103676a
More about Tellurium:
Imagine squeezing something in between two diamonds with a lot of force. What would it do to the thing? Well scientists did that with Tellurium and they found that it forms an atomic structure so complex that it doesn’t repeat itself. Tellurium is toxic and teratogenic (disruptive to the growth of a embryo or foetus), workers who exposed themselves to small quantities of Tellurium develop ‘tellurium breath’, which smells a lot like garlic. It was discovered in 1783 by a mine inspector named Franz-Joseph Müller von Reichenstein in Kleinschlatten, Transylvania. Müller was intrigued by ore from the mine as it had a metallic sheen. It was gold telluride, he studied the sample for 3 years and proved the presence of a new element. In 1789 he was sent another similar sample by a Hungarian scientist, Martin Kalproth, who then named it Tellurium. I wonder if they had garlic breath.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Tellurium in real crystal structures: