Sodium
Sodium:

Sodium burns with a bright yellow light. Sodium salts produce a gold or yellow color to fireworks. Public domain picture.
Facts about Sodium:
- Sodium: Soft, silvery metal at standard conditions
- Fun fact about Sodium: Although sodium is the sixth most abundant element on earth and comprises about 2.6% of the earth’s crust, it is highly reactive and it is never found free in nature!
- Chemical symbol: Na
- Atomic number: 11
A crystal structure containing Sodium:
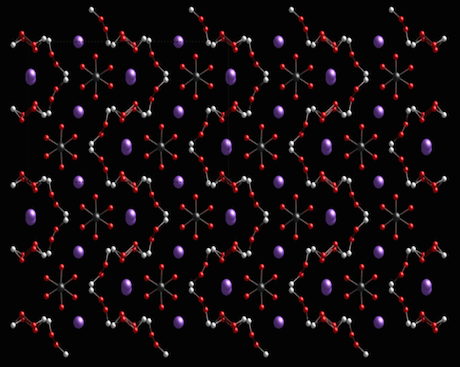
Ball-and-stick model of sodium percarbonate. Sodium is displayed as purple spheres. Attribution: Ben Mills/public domain picture.
Facts about this structure:
- Formula: 2(CO3 2-), 3(H2O2), 4(Na +)
- Structure name: tetrakis(sodium) tris(hydrogen peroxide) dicarbonate
- Fun fact about the structure: Sodium percarbonate is used as an environmentally-friendly bleach in cleaning products. Sodium percarbonate has Cmca symmetry at room temperature, while below 250K the structure changes to Pbca.
- CSD refcode: WUTKUT (What’s this?) This structure is also in the ICSD: 281382 (Find out more about the ICSD database)
- Associated publication: R. G. Pritchard, E. Islam, Acta Crystallographica,Section B: Structural Science, 2003, 59, 596, DOI: 10.1107/S0108768103012291
More about Sodium:
The name sodium derives from the English word soda because it was discovered in 1807 by the English chemist Humphry Davy from electrolysis of caustic soda (NaOH). The symbol Na derives from the Latin word natrium. The most common compound in which it occurs is sodium chloride (table salt). At room temperature, sodium metal is soft enough that it can be cut with a butter knife! Yellow street lamps often owe their color to sodium. Sodium-vapor lamps were invented in 1920 and use sodium in an excited state to produce light. Sodium is an essential element for all animals and some plants. More specifically, in animals, sodium is needed for the generation of nerve impulses and for preservation of electrolyte balance and fluid balance. Via the sodium-potassium pump, three sodium ions are pumped out of the cell and two potassium ions ions are pumped in.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Sodium in real crystal structures: