Oxygen
Oxygen:
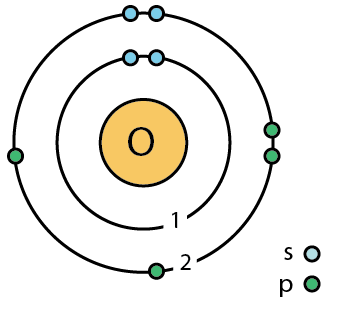
Image showing Bohr Model of Oxygen
Facts about Oxygen:
- Oxygen: Colourless, odourless and tasteless but when a liquid or a solid Oxygen is pale blue.
- Fun fact about Oxygen: Oxygen doesn’t like being lonely it always has a friend; two by two they endlessly roam the Earth. Sometimes Oxygen atoms run in packs of three and this is called ozone. Ozone is a thin layer of the atmosphere that protects us from the sun’s deadly UV rays.
- Chemical symbol: O
- Atomic number: 8
A crystal structure containing Oxygen:
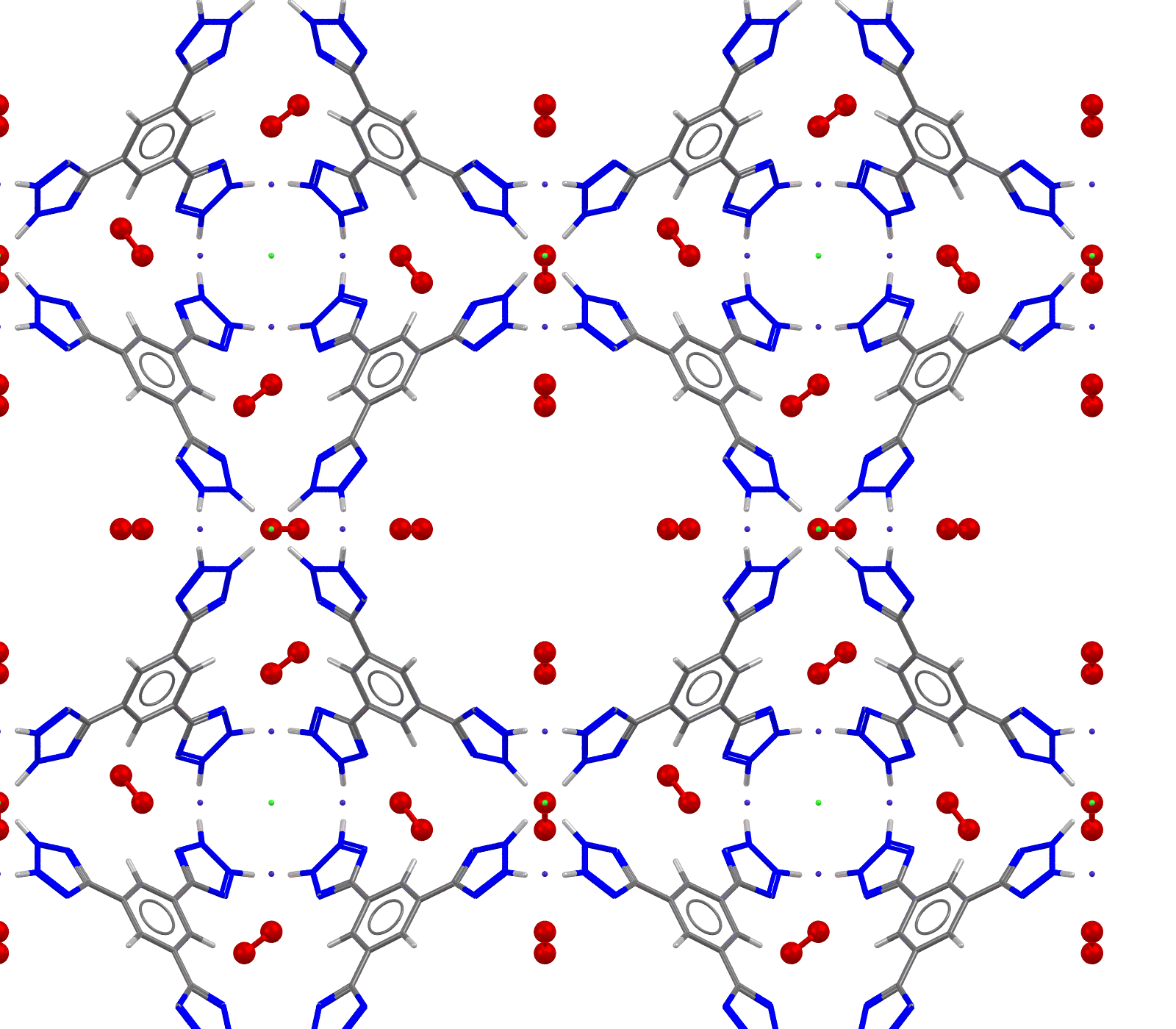
Image showing CSD Entry: AQOKUO, Oxygen atoms shown as red spheres.
Facts about this structure:
- Formula: 8(C13 H7 N8 3-),11.81(O2),13.5(Co 2+),3(Cl –)
- Structure name: cobalt(ii) (5,5′-[5-(pyrazol-1-id-4-yl)-1,3-phenylene]bis(1,2,3-triazol-1-ide)) chloride dioxygen
- Fun fact about the structure: IT LOOKS AMAZING!!! “The stability, selectivity, and high O2 adsorption capacity of these materials render them promising new adsorbents for air separation processes.”
- CSD refcode: AQOKUO (What’s this?)
- Associated publication: Dianne J. Xiao, Miguel I. Gonzalez, Lucy E. Darago, Konstantinos D. Vogiatzis, Emmanuel Haldoupis, Laura Gagliardi, Jeffrey R. Long, Journal of the American Chemical Society, 2016, 138, 7161, DOI: 10.1021/jacs.6b03680
More about Oxygen:
Oxygen is needed for you to stay living but if you inhale too much you can die… it’s not a fun way to go either. Let’s say you’re surrounded by air that contains 80% Oxygen, sounds okay, right? NOPE, your respiratory system will start getting irritated. With 12 hours of constant irritation and fluid build-up in your lungs, your end is near. Congratulations, now you can say you died by the very thing that was keeping you alive… but you can’t say that because you’ll be dead. This just proves that “too much of anything is bad”. Oxygen isn’t flammable but encourages the combustion of other flammable objects. Just think about it for a second… humans discovered fire a LOONNNNGG time ago, during the caveman era; surely by now the earth would have exploded with the amount of Oxygen in the atmosphere. Matches burn because Oxygen is the fuel to the fire. Surprisingly, we haven’t known about Oxygen for that long at all; we only discovered it in 1772 by a scientist who solved the age-old question – How do things burn?
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Oxygen in real crystal structures: