Nickel
Nickel:

Image showing a hard silver-coloured chunk of nickel metal, with the surface cut flat
Facts about Nickel:
- Nickel: Nickel is a metallic element with a silvery-white, shiny appearance at standard temperature and pressure (STP)
- Fun fact about Nickel: Nickel is magnetic at room temperature. (ref: Nickel Institute)
- Chemical symbol: Ni
- Atomic number: 28
A crystal structure containing Nickel:
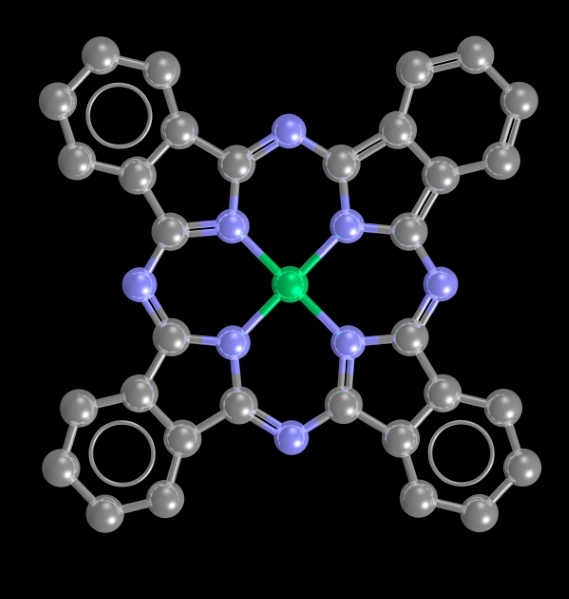
Image showing a structure containing nickel. Nickel is the central atom shown in green.
Facts about this structure:
- Formula: C32H16N8Ni
- Structure name: (phthalocyaninato)-nickel(ii)
- Fun fact about the structure: This structure was the first nickel-containing structure that was added to the CSD with 3D coordinates.
- CSD refcode: NIPHTC (What’s this?)
- Associated publication: J. Monteath Robertson, Ida Woodward, Journal of the Chemical Society, 1937, 219, DOI: 10.1039/JR9370000219
More about Nickel:
Nickel is the 5th most common element on the Earth with the concentration of nickel in the Earth’s crust being 80 parts per million. More than two thirds of the global nickel production is used to produce stainless steel which is useful in many different applications. Nickel(ii) phthalocyanine has the appearance of dark blue / purple powder or crystals and is used in thin film technology which has applications in gas sensing and electronic devices.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Nickel in real crystal structures: