Mercury
Mercury:

Image of the planet Mercury. Both the element and planet were named after the Roman God Mercury.
Facts about Mercury:
- Mercury: Silvery metallic liquid at standard conditions.
- Fun fact about Mercury: Although mercury and all of its compounds are known to be highly toxic, it was considered therapeutic throughout much of history!
- Chemical symbol: Hg
- Atomic number: 80
A crystal structure containing Mercury:
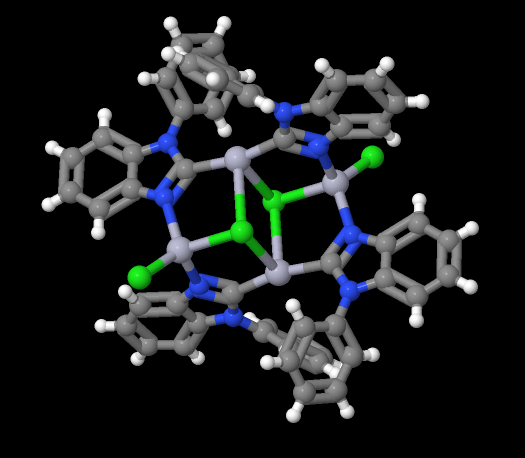
Image showing mercury atoms, displayed as light purple dots, in an anionic complex.
Facts about this structure:
- Formula: C52 H36 Cl4 Hg4 N8,C3 H6 O
- Structure name: tetrakis(μ-1-phenyl-1H-benzimidazol-2-yl)-bis(μ-chloro)-bis(chloro)-tetra-mercury(ii) acetone solvate
- Fun fact about the structure: This structure was obtained by a unique method to synthesize mercuramacrocycles.
- CSD refcode: MIXQES (What’s this?)
- Associated publication: Ming-Hung Yu, Hsueh-Hui Yang, Yu-Chen Gu, Bo-Han Wang, Fu-Chen Liu, Ivan J.B. Lin, Gene-Hsiang Lee, Journal of Organometallic Chemistry, 2019, 887, 12, DOI: 10.1016/j.jorganchem.2019.02.015
More about Mercury:
Mercury’s symbol Hg comes from its Greek name, hydrargyrum, meaning “liquid silver”, due to its mobility and shiny surface. Mercury is used in electrical switches of thermostats, but it is most commonly seen in the new the compact fluorescent light bulbs. Interestingly, mercury nitrate played a role in the term “mad as a hatter”. As mercury was used in the making of hats from animal pelts in the 18th century as it was used to clean the pelts before they turned them into hats. It was then discovered that a large percentage of the hatters working with it ended up suffering from brain damage due to mercury exposure. Mercury was found in Egyptian tombs that date back to 1500 BC. Amazingly, in China and Tibet, it was thought to prolong life, heal fractures, and maintain generally good health, although it is now known that exposure to mercury vapour leads to serious adverse health effects.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Mercury in real crystal structures: