Lithium
Lithium:
An Image of the Salar de Uyuni in Bolivia, the largest salt flat in the world. It is thought to contain one of the world’s biggest reserves of Lithium, which is contained in a Lithium-rich brine below the surface. Attribution: Lion Hirth (User:Prissantenbär) / Public domain
Facts about Lithium:
- Lithium: A silvery white solid
- Fun fact about Lithium: 7up, a popular soft drink in many parts of the world, used to contain Lithium! When it was first released in 1929 it was called “Bib-Labelled Lithiated Lemon-Lime Soda” and marketed as a health drink. Lithium (in the form of lithium citrate) was added to some soft drinks until the US government banned the use in the 1940s.
- Chemical symbol: Li
- Atomic number: 3
A crystal structure containing Lithium:
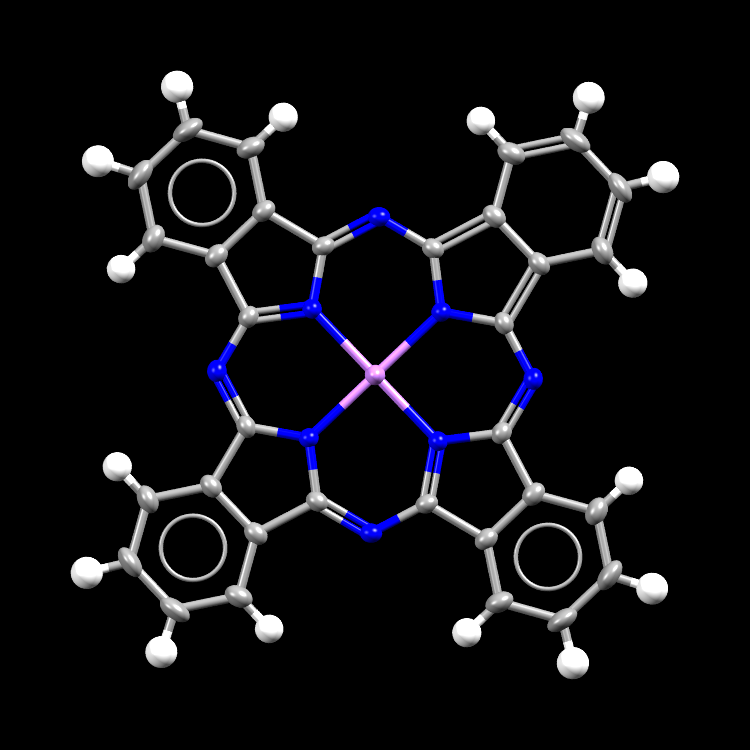
An image of a molecule of lithium phthalocyanine. Lithium is the central (purple) atom in the molecule.
Facts about this structure:
- Formula: C32 H16 Li N82
- Structure name: (Phthalocyaninanto radical)-lithium
- Fun fact about the structure: Lithium phthalocyanine is a semiconductor – a material which can conduct electricity at a rate in between the value for a conductor and an insulator. In the article, chlorine was added to the structure (a process called ‘doping’) to increase the conducting properties of the material.
- CSD refcode: DIXNIH03 (What’s this?)
- Associated publication: Yasuhito Miyoshi, Hirofumi Yoshikawa, Kunio Awaga, CrystEngComm, 2014, 16, 9266, DOI: 10.1039/C4CE01222D
More about Lithium:
Lithium was first discovered in the early 1800s and was named after the Greek word ‘lithos’ which means stone. It is a highly reactive metal and must be stored in oil to prevent it from reacting with air or moisture. Lithium can be extracted from some naturally occurring minerals or from the evaporation of lithium-containing brine (water with a very high salt concentration). It is has a low density – allowing it to float on water.
Lithium is used extensively in batteries. The Nobel prize in 2019 was awarded to John Goodenough, Stanley Whittingham and Akira Yoshino for their work in developing lithium-ion batteries. Lithium-ion batteries are rechargeable and are used in many devices, such as mobile phones and laptops. Lithium is an important component in the battery – the movement of lithium within the battery is part of the process that allows current to flow, when it is in a circuit.
Lithium also has a number of other applications. Lithium salts absorb carbon dioxide in air purification systems, including those used on submarines and spacecraft. Organolithium compounds are widely employed as reagents in chemical reactions. Lithium is also used as a red colourant in fireworks and in medicine as a drug to treat certain mental illnesses.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Lithium in real crystal structures:
