Francium
Francium:

Francium is expected to be reactive with water, much like this sodium explosion
Facts about Francium:
- Francium: Alkali metal, extremely reactive with water, extremely radioactive
- Fun fact about Francium: Francium only exists as a decay product of other elements and has a half life of at most 22 minutes
- Chemical symbol: Fr
- Atomic number: 87
A crystal structure celebrating Francium:
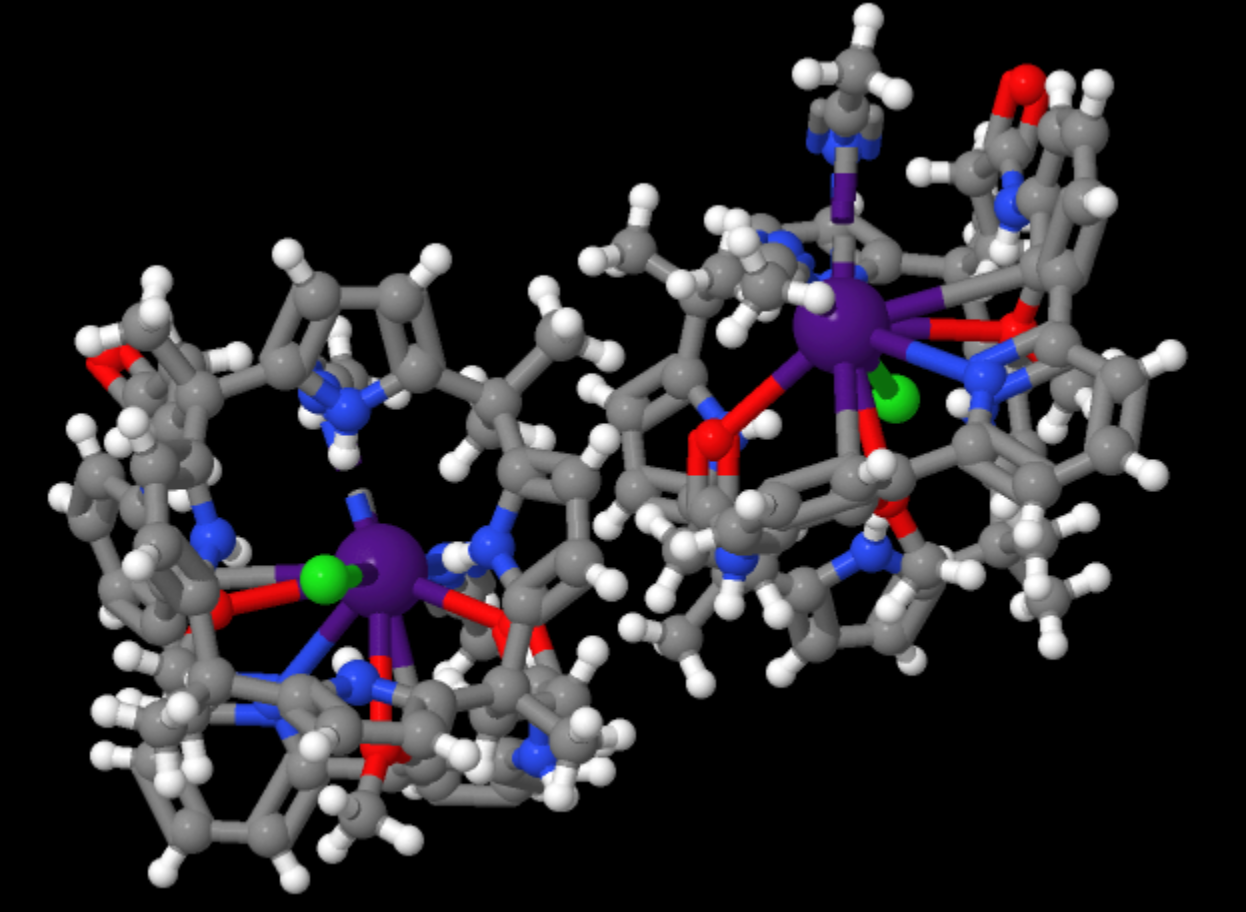
Structure showing how the organic extraction reagent binds Cesium cations
Facts about this structure:
- Formula: C55H61ClCsN9O4
- Structure name: bis(acetonitrile)-chloro-[48,50-bis(methoxy)-1,25,30,30,39,39-hexamethyl-5,21,44,45,46,47,49-heptaazanonacyclo[23.9.9.16,10.111,15.116,20.126,29.131,34.135,38.140,43]pentaconta-6(50),7,9,11(49),12,14,16(48),17,19,26,28,31,33,35,37,40,42-heptadecaene-4,22-dione]-cesium
- Fun fact about the structure: Francium along with other elements such as sodium and cesium are known as alkali metals. Due to its increased size Cesium is bound 10 times more weakly in this structure than Sodium would be.
- CSD refcode: QIDJEV (What’s this?)
- Associated publication: Qing He, Neil J. Williams, Ju Hyun Oh, Vincent M. Lynch, Sung Kuk Kim, Bruce A. Moye, Jonathan L. Sessler, Angewandte Chemie, International Edition, 2018, 57, 11924, DOI: 10.1002/anie.201805127
More about Francium:
Lithium is an essential element in almost all modern electronic devices, primarily in rechargeable batteries. However, recovering Lithium from devices is extremely difficult and only 1% is recycled world wide. This paper developed organic extraction reagents that were able to separate Lithium cations from other mixed alkali metal salts, providing a possible route to recycle lithium waste. By tuning the structure of the extraction reagent they could also tune which of the alkali metals they could extract. Francium is in the same elemental family as lithium and is expected to behave like other alkali metals. As a result you could design a similar structure that purifies Francium or leaves it in the mixture as desired.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Francium in real crystal structures: