Dubnium
Dubnium:

An impression of how superheavy elements are created; a small ion is made to collide with a large target atom with the aim of them combining to form a heavier element.
Facts about Dubnium:
- Dubnium: Dubnium is a synthetic element so its properties are unknown, but it is expected to behave like niobium and tantalum
- Fun fact about Dubnium: It took over 25 years to agree the name of element 105 as dubnium due to debate over who was the first to discover it. Other proposed names include nielsbohrium, hahnium and joliotium
- Chemical symbol: Db
- Atomic number: 105
A crystal structure celebrating Dubnium:
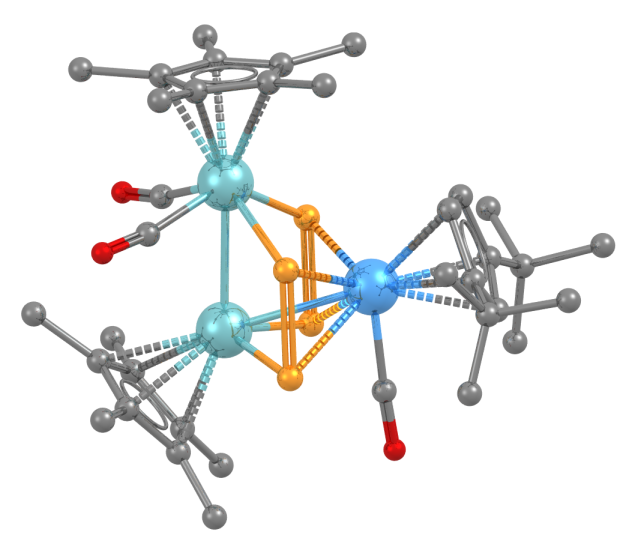
The structure has two niobium atoms (pale blue) and one tantalum atom (darker blue) linked by diphosphorus ligands.
Facts about this structure:
- Formula: C36 H51 Nb2 O3 P4 Ta
- Structure name: bis((η5-Pentamethyl-cyclopentadienyl)-(μ2-η2,σ2-diphosphorus))-(η5-1,3-di-t-butyl-cyclopentadienyl)-tricarbonyl-di-niobium-tantalum
- Fun fact about the structure: The structure is one of only two in the CSD to contain the group 5 elements niobium and tantalum together – dubnium should behave in a similar way to these metals. If it was possible to make a structure containing dubnium it might look something like this!
- CSD refcode: WAKBOZ (What’s this?)
- Associated publication: O.J.Scherer, R.Winter, G.Wolmershauser, Zeitschrift fur Anorganische und Allgemeine Chemie, 1993, 619, 827, DOI: 10.1002/zaac.19936190504
More about Dubnium:
Dubnium is a superheavy synthetic element that does not occur in nature. The Joint Institute for Nuclear Research in Dubna, Russia, first claimed to have discovered the element in 1968; the element was formally named dubnium after the town of Dubna in 1997. As with all of the superheavy synthetic elements, dubnium is highly radioactive and unstable, meaning that opportunities for research involving the element are very limited. However, as the longest-lived isotopes of dubnium have a half-life of about 1 day, some research into properties of dubnium has been carried out. These studies suggest the element behaves most like niobium. The study of superheavy elements like this is useful as it gives an insight into why some nuclei are more stable than others.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Dubnium in real crystal structures: