CSD Educators: Anthony L. Fernandez from Merrimack College, US
As part of the CSD Educators series we are proud to feature a special introduction from CCDC fellow, Greg Ferrence, on a blog written by Anthony L. Fernandez.
Introduction from Greg Ferrence during a CCDC sabbatical:
It is my pleasure to welcome Professor Anthony Fernandez as our guest contributor to this blog in the CSD Educators series. Anthony is Professor and Chair of Merrimack College’s Department of Chemistry and Biochemistry. (In fact, he was recently promoted to Full Professor; congratulations Dr. Fernandez!) Anthony was educated as an inorganic chemist and earned his Ph.D. from Boston University. It was later, during his first sabbatical, that he discovered for himself, the pleasures and powers of SCXRD and the CSD. He soon became an advocate and innovator in the use of the CSD in Chemistry Education. In addition to developing a handy spreadsheet indexing the CSD Teaching Subset, he has developed and continues to develop learning objects that incorporate data from the CSD. He makes these resources freely available to other educators via the web-based Virtual Inorganic Pedagogical Electronic Resource (https://www.ionicviper.org). Anthony, the mic… err… blog is yours.
From Anthony L. Fernandez from Merrimack College, US
My first structure
For many years, I felt like an impostor—I was an inorganic chemist who had never had a crystal structure determined for a compound I had made. I did an entire PhD, a postdoctoral fellowship, and several years of a tenure-track faculty position without growing an x-ray quality crystal. During my first sabbatical, I worked with Professors Oleg Ozerov and Bruce Foxman at Brandeis University and acquired several crystal structures with their new diffractometer. (The first crystal structure that I acquired, BESDIN01, is shown below.[1]) Bruce helped me through the process and to think about how to use the information gleaned from solid-state structures could be used to explain the observed chemistry. This experience also caused me to ask how I could use crystal structures in my teaching. For many years, I had drawn structures in chemical structure-drawing programs and I was disappointed that my students were not getting a chance to investigate real structural data on their own.
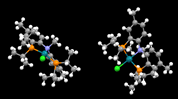
The first crystal structure that I acquired, BESDIN01
Incorporating structural data into university courses
While thinking about how to incorporate solid-state structures into my courses, I read a number of articles by Battle, Ferrence, and Allen about how to use structural data in education. I began to use Mercury to investigate the Teaching Subset of the Cambridge Structural Database (CSD) and quickly realized that a detailed index of the structures in the subset would be useful. I started by keeping track of the structures that I used in my inorganic chemistry class on index cards. When I got up to about 50 cards, I created a spreadsheet that contained information (metal, coordination number, ligands, etc) about these structures. As this spreadsheet then grew to contain information about all of the metal-containing structures in the Teaching Subset, I realized it would be helpful to other users of the CSD Teaching Subset and to those who teach inorganic chemistry, so I took the opportunity to make it available on the Virtual Inorganic Pedagogical Electronic Resource (VIPEr) website (https://www.ionicviper.org/) as a Learning Object (“Annotated List of Metal-Containing Structures in the Cambridge Structural Database Teaching Subset”).[1] The Interactive Online Network of Inorganic Chemists (IONiC) created the VIPEr website to be a place where faculty can share inorganic chemistry teaching materials and build an online community by collaborating with others to improve the teaching of inorganic chemistry.
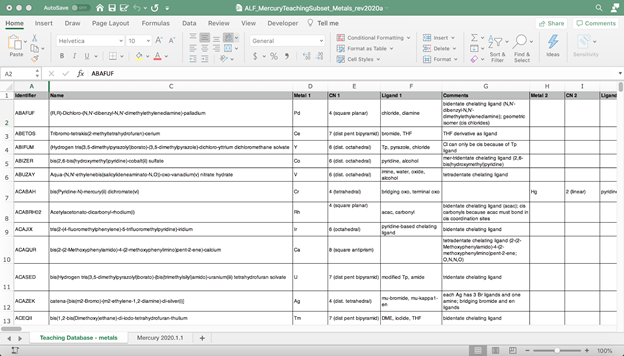 Image showing details of metal-organic structures in the CSD Teaching Database
Image showing details of metal-organic structures in the CSD Teaching Database
Shortly thereafter, I met Amy Sarjeant, who, at the time, was the CCDC’s Outreach and Education Manager, at an ACS conference and we talked at length about how I use the Teaching Subset in my courses. I shared a number of activities that I had created and she encouraged me to think about what other structures could be added to the Teaching Subset to make it more useful. I suggested a number of structures for addition so that there were examples of all of the geometries included in inorganic textbooks, different forms of several linkage isomers, and complexes that illustrated the impact of metal oxidation state on metal-ligand bond length.
Linking structural data with experiments
One of my favorite experiments focuses on the synthesis of metal-DMSO complexes, in which Students make DMSO complexes of several metals (Cu, Pd, Sn, Ru) and use infrared spectroscopy to determine the binding mode (S- or O-bonded) of the DMSO ligand. The IR peak for the S-O vibration shifts according to binding more, wth O-bonded ligands having a higher vS-O and S-bonded ligands having a lower vS-O than that for free DMSO. When drawing the resonance structures that are used to explain this change in the infrared spectra, I wondered if this would be visible in the crystal structures of these complexes. While this could be seen in the Rh(III) complexes of DMSO (IMOCAP, see below) that I used in my pre-lab assignment[1], I was curious if this was a more general phenomenon.
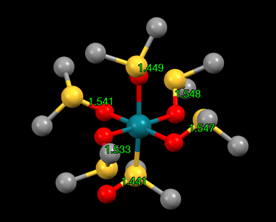
An Rh(III) complex of DMSO (CSD Refcode IMOCAP)
At this point, I did not have access to the full version of the CSD and I had to turn to my friend, Bruce Foxman, to help me with the investigation. He showed me how to search for this data in ConQuest and an analysis of the results showed that the O-bonded DMSO ligands had longer S-O bond lengths, on average, than S-bonded DMSO ligands. I was excited to see these results and it led me to work with our library to get a subscription to the full CSD so that I could expand the use of the CSD and ConQuest in my teaching. My department and the library currently share the cost for a license that allows us to have the CSD Suite installed on one computer in the department and one computer in the library so that any faculty member or student can access this resource. Over the past several years, I have been encouraging my colleagues to think about how the CSD can be used to enhance their teaching and support their research.
A move to more virtual learning
This year, given the COVID restrictions on in-person research activities on campus , I have started to use the CSD more broadly in my research. Last semester, my research student and I used the FindGeo tool[1] (implemented as part of the MetalPDB website) to determine the coordination geometry of all of the metal-containing structures in the teaching subset. My student and I saved each metal-containing structure from the Teaching Subset as a PDB file so that we could import it into the FindGeo tool and calculate the best geometry.
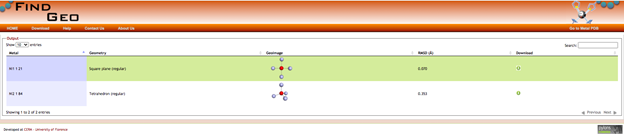 The FindGeo tool
The FindGeo tool
We identified the most common metal coordination geometries, which were octahedral and tetrahedral (to no one’s surprise), and confirmed the geometric assignments for structures with coordination numbers greater than 7. We also found that a number of structures (those with a CN > 9 or those with olefins or Cp rings as ligands) could not be analyzed using this tool. This result led us to ask how we could determine the geometry of any structure containing a π-bonded olefin ligand, which includes those with η5-cycpentadienyl, η6-arene, η2-olefin, and other related ligands. Do we include all of the atoms of the π-bonded ligand or should we just use the centroid of the ligand when assigning the geometry of the complex? We are currently searching for complexes with a variety of pi-bonded ligands in the full CSD using Conquest. The centroid of the π-bonded ligand (determined as part of a 3D parameter in ConQuest) is used as a reference point so that the angles between the ligands can be measured and the “geometry” of the complex can be determined. We have gotten some really interesting preliminary results and my research student is continuing to search for other types of π-bonded olefin ligands and analyzing the results of her searches.
Given all of the excellent experiences that I have had so far with the CSD and the Teaching Subset, I look forward to expanding my capabilities so that I can find new ways to support my research efforts and further improve my teaching of structural inorganic chemistry.
References
1 Fernandez, A.L.; Ozerov, O.V.; Foxman, B.M. CCDC 2015360: CSD Communication, 2020. DOI: 10.5517/ccdc.csd.cc25n4mm
3 Fernandez, A.L. DMSO as a ligand in coordination chemistry. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2018. {Problem Set} https://www.ionicviper.org/problem-set/dmso-ligand-coordination-chemistry (accessed May 18, 2018)
4 Andreini, C.; Cavallaro,G.; Lorenzini, S. FindGeo: a tool for determining metal coordination geometry.
Bioinformatics 2012, 28, 1658-1660. http://metalweb.cerm.unifi.it/tools/findgeo/
A list of my other Learning Objects that incorporate data from the Teaching Subset or the full CSD
- Fernandez, A.L. First Look at Coordination Complexes. Virtual Inorganic Pedagogical Electronic Resource, [Online] September 2020. {In-Class Activity} https://www.ionicviper.org/class-activity/first-look-coordination-complexes (accessed October 20, 2020)
- Fernandez, A.L. Geometry Indices. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2018. {Five Slides About} https://www.ionicviper.org/five-slides-about/geometry-indices (accessed August 22, 2018)
- Fernandez, A.L. Isomerism in Coordination Complexes. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2018. {In-Class Activity} https://www.ionicviper.org/class-activity/isomerism-coordination-complexes (accessed July 9, 2018)
- Fernandez, A.L. Metal Tropocoronand Complexes. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2018. {In-Class Activity} https://www.ionicviper.org/class-activity/metal-tropocoronand-complexes (accessed May 18, 2018)
- Fernandez, A.L. DMSO as a ligand in coordination chemistry. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2018. {Problem Set} https://www.ionicviper.org/problem-set/dmso-ligand-coordination-chemistry (accessed May 18, 2018)
- Fernandez, A.L. Annotated List of Metal-Containing Structures in the Cambridge Structural Database Teaching Subset. Virtual Inorganic Pedagogical Electronic Resource, [Online] August 2015. {Web Resources and Apps} https://www.ionicviper.org/web-resources-and-apps/annotated-list-metal-containing-structures-cambridge-structural-database
- Fernandez, A.L. Introduction to Mercury. Virtual Inorganic Pedagogical Electronic Resource, [Online] January 2015. {In-Class Activity}
- https://www.ionicviper.org/class-activity/introduction-mercury (accessed May 18, 2018)