Thallium
Thallium:

Image of Hutchinsonite, a common mineral containing thallium in the crust.
Facts about Thallium:
- Thallium: Solid, dull metallic colour, tasteless, air sensitive.
- Fun fact about Thallium: Soluble Thallium salts were common in insect and rat poisons as well as murder weapons due to their toxicity.
- Chemical symbol: Tl
- Atomic number: 81
A crystal structure containing Thallium:
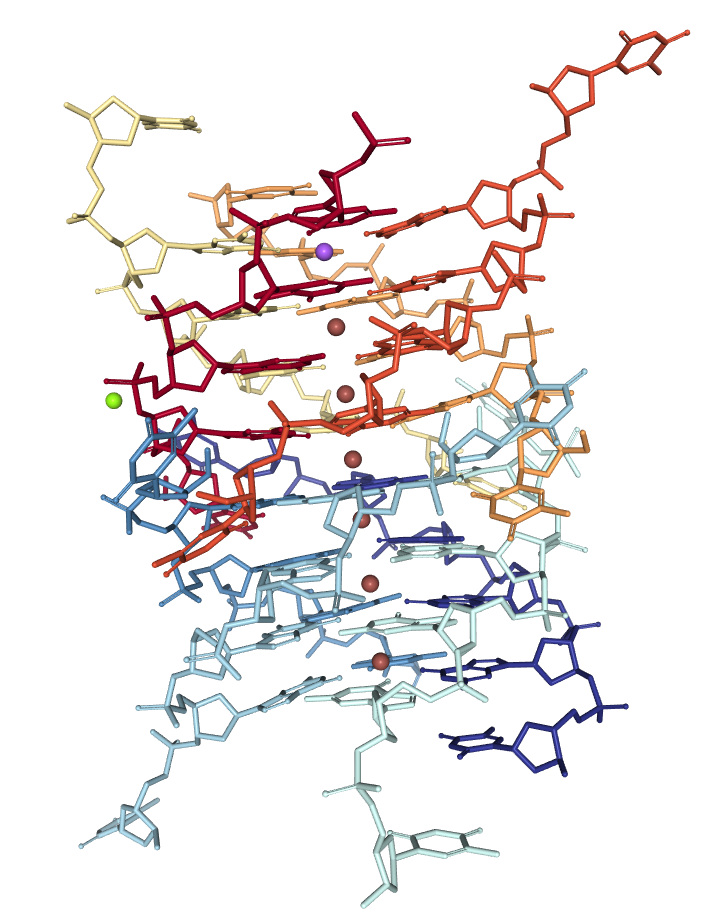
Image showing a G-rich strand of DNA stabilised into a Quadruplex structure stabilised by Tl+ ions
Facts about this structure:
- Formula: d(TGGGT)4 Na2 Tl5 Mg1 (H2O)112
- Structure name: S1 d(TGGGT) Quadruplex
- Fun fact about the structure: Thallium is the only group III element that stabilises DNA quadruplexes.
- PDB code: 1S45 (More info about the PDB)
- Associated publication: Carmen Cáceres, Glenford Wright, Catherine Gouyette, Gary Parkinson, Juan A . Subirana, Nucleic Acids Research, 2004, 32, 1097, DOI: 10.1093/nar/gkh269
More about Thallium:
Thallium is a relatively rare element in our day to day lives, but has been shown to displace other cations in biological contexts such as DNA as well as binding to the sulfur found on cysteine residues in proteins. This makes Thallium salts extremely toxic. Heavy elements are often used to help crystalise biological and structures. Despite Thallium being in a very different part of the periodic table, its similar size and charge to potassium salts means it can fulfil the same role in natural quadruplex structures. These quadruplex structures are implicated in both normal and cancer biology and are of interest to researchers around the globe.
Thallium isn’t just present in protein structures like DNA, it can also be found in small molecule structures in the CSD for example CSD Entry WIKQUC which is described in DOI: 10.1021/ic991492n. This compound displays a strong visible luminescence, under UV excitation, which is sensitive to temperature due to the interaction the Thallium ion has with gold.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Thallium in a real crystal structure: