Rhenium
Rhenium:
A high purity (99.999 %) rhenium single crystal made by the floating zone process, an ebeam remelted (99.995 %) rhenium bar and as well as a high purity (99.99 % = 4N) 1 cm3 rhenium cube for comparison. Attribution: Alchemist-hp (talk) (www.pse-mendelejew.de) [FAL]
Facts about Rhenium:
- Rhenium: White silver-white metal, has the third highest melting point of any of the metals only exceeded by tungsten and carbon. It is extremely dense the only elements that exceed it are platinum, iridium and osmium.
- Fun fact about Rhenium: It is the last stable, non-radioactive, naturally occurring element that has been discovered.
- Chemical symbol: Re
- Atomic number: 75
A crystal structure containing Rhenium:
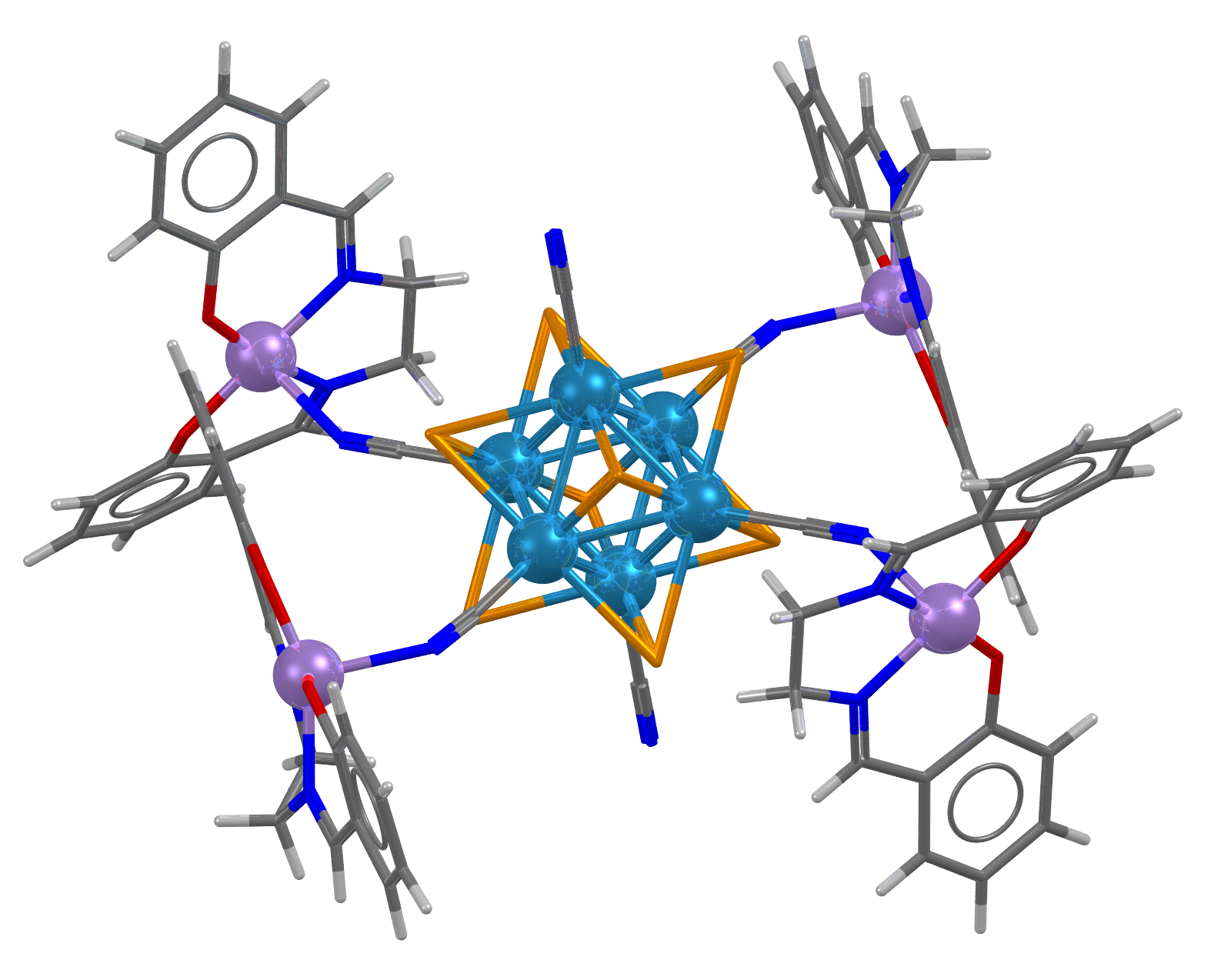
Image showing CSD Entry: ACAHAN, Rhenium atoms shown as blue spheres.
Facts about this structure:
- Formula: (C70 H56 Mn4 N14 O8 Re6 Te8)n
- Structure name: catena-(octakis(μ3-Tellurido)-hexakis(μ2-cyano-C,N)-tetrakis(N,N’-bis(salicylidene)ethane-1,2-diamine-N,N’,O,O’)-tetra-manganese(iii)-hexa-rhenium)
- Fun fact about the structure: This cyano bridged mixed metal cluster is a 2D layered extended network structure.
- CSD refcode: ACAHAN (What’s this?)
- Associated publication: Youngmee Kim, Seon-Mi Park, Wonwoo Nam, Sung-Jin Kim, Chemical Communications, 2001, 1470, DOI: 10.1039/b104276a
More about Rhenium:
In 1925, the German Chemists Walker Noddack, Ida Tacke and Otto Berg isolated Rhenium from platinum ores. The Russian Chemist Dmitri Mendeleev predicted there would be an element closely related to manganese, he called this‘div-manganese’. This was the first prediction of the existence of Rhenium.
Have you ever used an oven? If so, you have utilised the ultra-high melting point of Rhenium, at a whopping 3453K (3179.85°C). Rhenium is used as an additive to tungsten and molybdenum-based alloys (also elements which have high melting points) to increase the ductility (allows it to withstand stress). This metal despite its rarity is commonly found in almost all electric ovens, filament lamps, even if it is a miniscule amount. It is even used with platinum as a catalyst in the production of high octane, lead-free gasoline.
The same stuff is used in the production of jet engines; nickel-based superalloys containing 6% Rhenium are used to make the combustion chambers, turbine blades and exhaust nozzles of jet engines. F-22 Raptors and F-35 aircrafts all contain this superalloy.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Rhenium in real crystal structures:

.jpg)
