Polonium
Polonium:

A thin film of polonium on a stainless-steel disk. Picture credit: Albert Fenn, taken from the LIFE science library.
Facts about Polonium:
- Polonium: Solid and metallic at room temperature
- Fun fact about Polonium: Polonium is a rare and radioactive elemement. It was discovered by Marie Curie, who named it after her homeland, Poland.
- Chemical symbol: Po
- Atomic number: 84
A crystal structure containing Polonium:
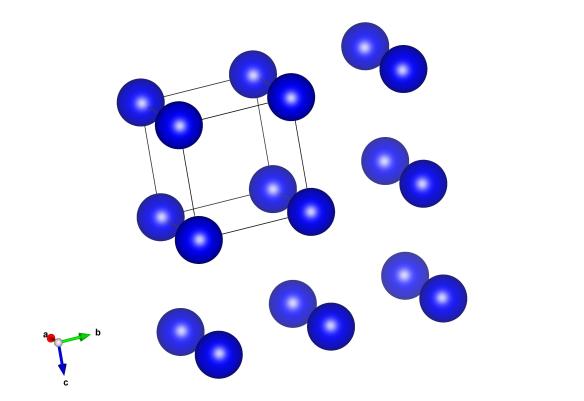
Image shows the simple cubic structure of Polonium
Facts about this structure:
- Formula: Po
- Structure name:Polonium
- Fun fact about the structure: Though it looks very simple, this crystal structure of polonium is a bit of a strange one. First discovered by Marie and Pierre Curie in 1898, It is the only element to form into a simple cubic structure, a cube with an atom sitting at each corner.
- ICSD number: 43211 (Find out more about the ICSD database)
- Associated publication: R.J. Desando, R.C. Lange, Journal of Inorganic and Nuclear Chemistry, 1966, 28, 1837, DOI: 10.1016/0022-1902(66)80270-1
More about Polonium:
How this very simple crystal structure stacks up is that each of the Polonium atoms sit on top of each other. Don’t you think this is that odd? Try taking some ping pong balls – and then construct the next layer so that each ball in this new layer sits directly on top of the one below. It’s not very stable is it? All other elements form ‘close packed’ structures, like Gold (cubic close packing) and Krypton (hexagonal close packing). In these structures the next layer sits in the dimple left between atoms in the layer below. Polonium, it would seem, is very different. This very simple (but strange) structure was first described in 1966, along with how the structure can change when you heat it up. More recently a group of theorist have investigated why Polonium takes up such a simple structure.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Polonium in real crystal structures: