Manganese
Manganese:
Manganese oxide dendrites on a limestone bedding plane from Solnhofen, Germany. Scale in mm Picture credit: Mark A. Wilson (Department of Geology, The College of Wooster)
Facts about Manganese:
- Manganese: Solid and metallic at room temperature, silvery grey and looks a bit like iron.
- Fun fact about Manganese: Some of the oxides of Manganese can be very colourful, and found abundantly in nature. Many historic cave paintings, such as the ones in Lascaux, France include pigments that are Manganese oxides.
- Chemical symbol: Mn
- Atomic number: 25
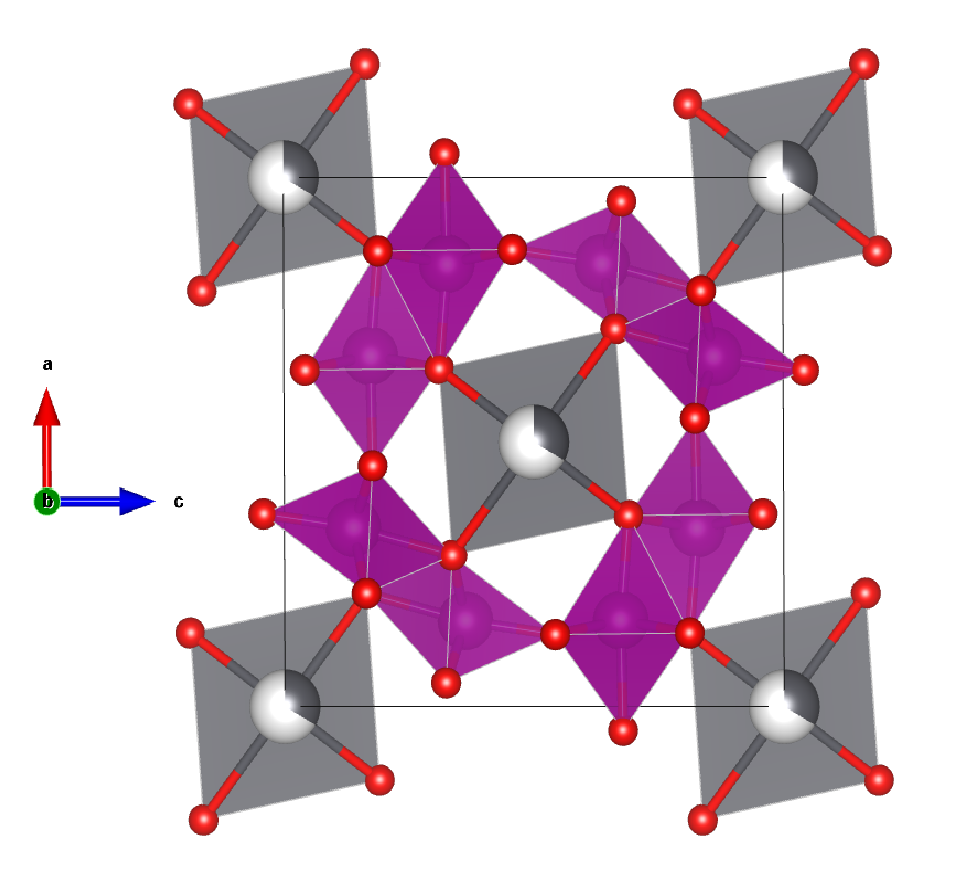
A crystal structure containing Manganese:
Image shows the crystal structure of the mineral Coronadite
Facts about this structure:
- Formula: Mn8 O16 Pb1.34
- Structure name: Lead manganese oxide, or mineral name Coronadite
- Fun fact about the structure: How can something inorganic look organic? When it is a Manganese oxide. There are many cave systems where you will see traces like those in the picture, but they are a moss or lichen growing in cracks, not a mineral. Manganese oxides, often in their hollandite structure form (like that featured) are picked up by fluids and get deposited, but do look a bit like they are living.
- ICSD Number: 203079 (Find out more about the ICSD database)
- Associated publication: J.E. Post, D.L. Bish, American Mineralogist, 1989, 74, 913, Link to publication
More about Manganese:
Manganese has a very close relationship with iron, in fact they are next to each other in the periodic table! This also goes back to when the Greeks first described Manganese, two materials had been discovered in the city of Magnesia, both called ‘magnes’ but separated by the masculine or feminine form of the word. The masculine one was known to attract iron and the feminine one was use to colour glass. As we now know that Manganese isn’t magnetic, then the feminine form of magnes is thought to have been a Manganese oxide (with the masculine one being an iron oxide). Manganese continues to be vital for steel production, as it is able to help remove impurities such as oxygen and sulfur. So next time you cross a steel-braced bridge, you can thank Manganese for keeping it strong for you. As well as forming minerals like Coronadite, Manganese is also present in other metal-organic structures. The structure of a nano-sized magnetic molecular wheel containing Manganese (CSD Entry: VIVCOV) is displayed in the visualiser below.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Manganese in real crystal structures: