Californium
Californium:

Image of Californium which is a silvery-white radioactive metal
Facts about Californium:
- Californium: Silvery-white metal, slowly tarnishes in air at room temperature. Two crystalline forms exist under normal pressure whilst the third exist under high pressure.
- Fun fact about Californium: Californium is a malleable and so soft a metal that it can be sliced with a razor blade. Its spectrum has been observed in supernova. 22 g of Californium cost 22 million pounds-one of the most expensive substances in the world.
- Chemical symbol: Cf
- Atomic number: 98
A crystal structure containing Californium:
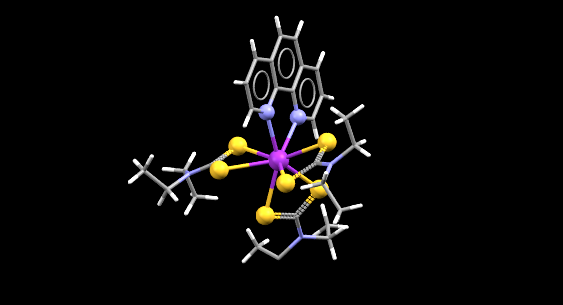
A ball and stick representation of a Californium complex (Cf depicted as the purple ball)
Facts about this structure:
- Formula: C27 H38 Cf N5 S6,C2 H3 N
- Structure name: tris(diethylcarbamodithioato)-(1,10-phenanthroline)-californium acetonitrile solvate
- Fun fact about the structure: A crystal structure of this complex suitable for single x-ray diffraction was grown just over the course of an hour.
- CSD refcode: FIHLIU (What’s this?)
- Associated publication: Samantha K. Cary, Jing Su, Shane S. Galley, Thomas E. Albrecht-Schmitt, Enrique R. Batista, Maryline G. Ferrier, Stosh A. Kozimor, Veronika Mocko, Brian L. Scott, Cayla E. Van Alstine, Frankie D. White, Ping Yang, Dalton Transactions, 2018, 47, 14452, DOI: 10.1039/C8DT02658K
More about Californium:
Californium, named after the University of California, Berkeley, and the state of California is the heaviest element to be produced in weighable amounts. With a +3 dominant oxidation state (OS), it becomes only water soluble at this OS. Efforts to reduce or oxidize the +3 ion in solution has seen failure. The actinide element produced by bombarding curium with alpha particles has its electronic properties affected by the movement of its inner electrons at significant factions of the speed of light. The most stable laboratory-synthesized isotope Cf-252 has a half-life of approximately 800 years and is a very strong neutron emitter which means it possesses atypical property of giving off neutrons when it breaks apart and it is interesting because isotopes that behave in this way are very unusual. Neutrons from Californium (Cf-252) are utilized to serve the treatment of brain and cervical cancers in instances where radiation therapy proves futile.
Learn More About the International Year of the Periodic Table (IYPT) in Crystals Project:
This project (#IYPTCrystals) is part of the International Year of the Periodic Table celebration (#IYPT2019), read more about the project here.
You can follow us on social media; search for #IYPTCrystals or follow The CCDC on X @ccdc_cambridge on Facebook ccdc.cambridge, on Instagram ccdc_cambridge or on YouTube CCDCCambridge.
Understand some of the terms and concepts used with our Frequently Asked Questions page here.
A 3D visualization showing Californium in real crystal structures: