IYPT through Crystals: voices, elements and structures
Reflections on the IYPT in Crystals project - including the competition winners, educational resources you can use at home, and what's next for the fantastic entries collected from across the community.
The IYPT through Crystals project was launched in 2019 from the partnership between CCDC, the British Crystallographic Association (BCA) and the National Crystallography Service (NCS), and has since then continuously been growing.
I must say I joined the celebrations late, as I have been at CCDC for just over a month at the time of writing. And yes, you did count right: I started after the beginning of the lockdown in UK, so I have never met my colleagues in person, only via a screen! As the new Education and Outreach Officer, I was immediately involved in the IYPT through Crystals project, which for me was also the first opportunity to collaborate with the team. And what an exciting project to take part in!
Over the last month, we have seen many updates coming from this project: the periodic table of crystals is very close to be complete thanks to the contributions of the crystallographic community; we have just crowned the #IYPTCrystals champion (if you have not seen it on social media, the revelation of the winner is only few paragraphs away); and the new IYPT Crystals Battlecards are currently the most popular educational resource on our website. This blog is a chance for us to thank all the contributors to this project, catch up with the latest news, and look towards our next steps.
Frequently Asked Questions about the IPYT through crystals project
Thank you to our community volunteers!
Building an interactive periodic table of elements and crystals structures in their honour would have not been possible without a great team. And it took me a while to realise how big the team really is. While catching up with the exploration of our periodic table, I could not avoid noticing how many different voices and styles were emerging element after element. And indeed, it is a community-led project, with contributors from all over the world, who dedicated their time to curate one or more element pages. You can meet some of the authors in our contributors page, and here are some statistics. Fifty-eight volunteers took part in the project from 13 different countries.
Among the most prolific external contributors, we find Laura McCormick McPherson and Amy Sarjeant, who both curated 10 elements, Tanay Menon with 8, Rudra Narayan Samajdar and Claire Murray (who also pioneered this project) with 5 element pages, Samuel Roberts and Helen Maynard-Casely with 4 each, and Colin Edge with 3 elements. And we cannot forget in this list Gregory Ferrence, who involved his students in the project, and they curated 12 element pages overall.
We really want to take the opportunity in this blog to thank all the volunteers: thank you for making such an ambitious project possible!
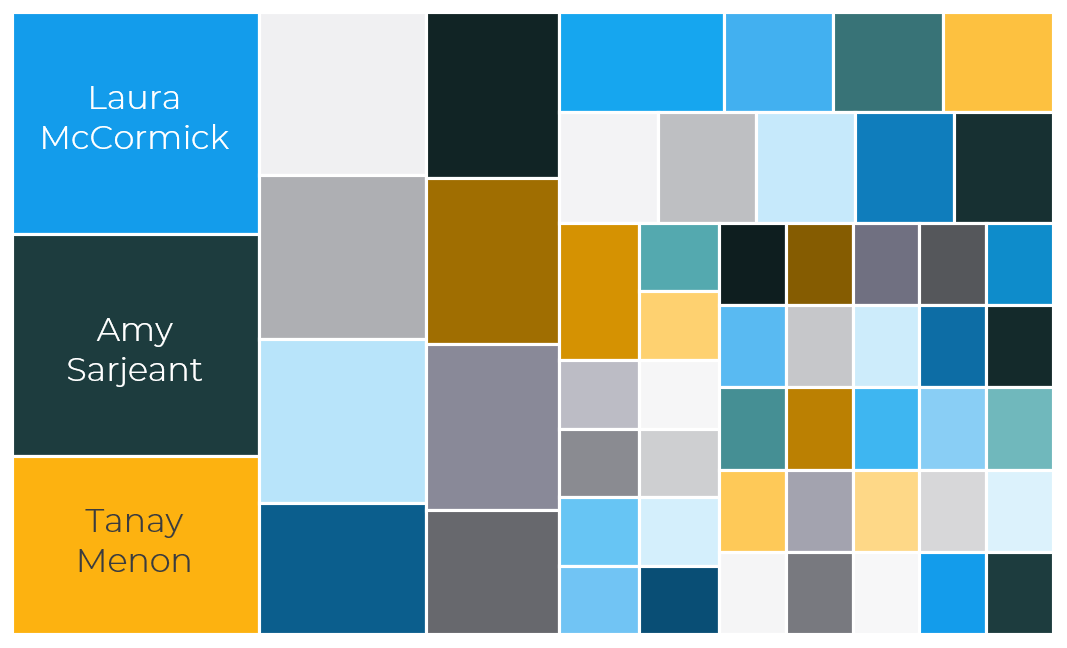
Each contributor (or team of contributors) is represented by a coloured square, with area proportional to the number of elements curated. The names of the top 3 contributors are also shown.
Competition – the public decides!
To reward the hard work in putting together the periodic table, on the 4th of May we launched the #IYPTCrystals competition, which will bring glory and a 3D structure of a crystal to the winner.
From all the elements, our panel of judges selected carbon, chromium, cobalt, europium, germanium, and tellurium to enter the final round of the competition, thanks to their highly engaging and informative webpages, and to the high number of views. But the final decision of the winner was in your hands and collectively you responded with over 2,000 votes.
We have been monitoring the votes behind the scenes and what a nail-biting finale with two elements at the top of the leader board with only few votes away from each other on the last day! So, finally, the winner is… Cobalt! Congratulations to Alexis Graybeal and Chiara Lagioia, authors of this elements page! I am glad to share with you what the authors told us about themselves and the making of cobalt’s webpage:
“Hello! Our names are Alexis Graybeal and Chiara Lagioia, the cocurators of the cobalt webpage. We are both students at Illinois State University studying chemistry and middle school education, respectively. We were involved in the IYPT Crystals project as an assignment for our university’s inorganic chemistry lab course. We chose to create the webpage for cobalt because we loved the color of the element and we had recently been studying transition metal complexes in our inorganic chemistry course. The crystal structure included in the cobalt webpage was refcode: AQOLAV. We chose this crystal structure because we thought it looked very unique. Thank you to everyone who voted, we are glad you enjoyed the page!”
Congratulations!

(Left) The final leader board of the elements that entered the final round of the #IYPTCrystals competition. (Right) The curators of cobalt, Chiara Lagioia (top) and Alexis Graybeal (side), in a frame of the structure they chose (AQOLAV).
Educational resources and future work
The competition for us was only the start of promoting engagement and learning with our periodic table of crystals. Indeed, the IYPT through Crystal team had the aim from the start to develop educational resources that could be used in schools, science festivals, and by families.
The collaboration with NCS was a great fit for this part of the project. Simon Coles, Director of the NCS, Education and Outreach Coordinator for the BCA, good friend of the CCDC, and part of the IYPT through Crystals team, has just the right words for this;
“This project was conceived at the ECM in Oviedo, along with Claire Murray from Diamond Light Source. Right from the outset this was an education-driven effort and the first thing we did to get it started was apply to the RSC to support the development of educational resources. This really helped to shape the way we went about developing the project. The periodic table as we know it is presented in its more technical form (for good reason!), but the general public and those in earlier stages of education don’t tend to have an easy or engaging way to understand it. By using everyday examples and the great interactive nature of 3D crystal structures I believe we have found a genuinely novel and interesting way to present it. The challenge now is to develop engaging education and outreach activities that build on this great resource – watch this space (or get in touch to get involved)!”
The first resource we have built that uses our periodic table of structures are the IYPT Crystals Battlecards. You can find this fun and educational game at this link. Each battlecard in the deck represents an element from the periodic table, from hydrogen to xenon, its celebrative crystal structure and their properties are the grounds for the game. These battlecards shall thus represent a window for children and players of any age and background on the crystallographic world and offer a fun way of learning while strategising for the victory. To encourage curiosity and creativity, we also provided empty cards templates, so everyone can include their own personalised cards in the deck, inspired by our periodic table of crystals or by their own favourite structures. And I would not be surprised to see structures yet to be discovered in the home-made cards, because what would scientific research be without the dream to create something that does not exist today?
What is next for the IYPT through Crystals project?
The team that never sleep (ok, that is just because we have team members in different time zones, so there is always someone awake) have a couple more ideas to keep the fun going. We are planning new resources to engage with the periodic table and structures from the CSD, and to encourage learning about elements and crystals. Once the activities will be ready, you will find them in our activities webpage and on our social media: watch these spaces!
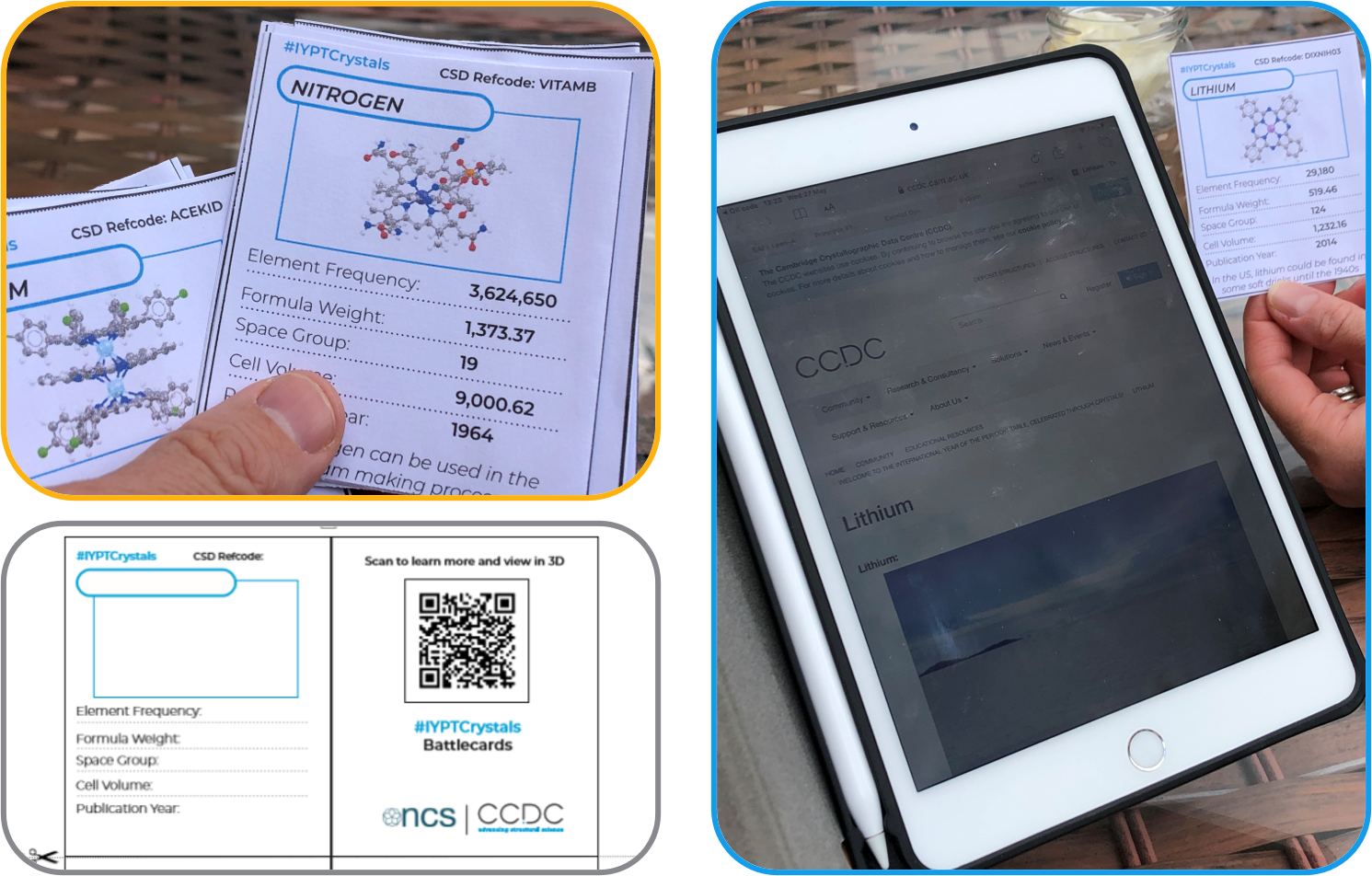
(Top and right) The Battlecards in action – thank you to Chris Tromans. (Bottom) Personalised cards template.
While the International Year of the Periodic Table has ended, as we are half-way into 2020, the IYPT Crystals project is still very much alive and will keep growing thanks to the voices of many contributors. We hope it will keep engaging and inspiring others to discover more about elements and crystal structures.